- Home
- Portfolio
- About
- Contact
- Blog
- The Bitcoin Standard Pdf
- 'LINK' Rexus Mouse Driver Downloadl
- Junooniyat Movie In Torrent Download yaffval
- Kundli 2012 Software Free Download !!TOP!! Full Version With Crack
- High Quality God_of_war_4_strategy_guide_pdf_
- Candydoll.TV - Laura B. - Sets 1-30
- Candydoll Valensiya Set 31 [WORK]
- [VERIFIED] Soal Ujian Diklat Pim 4 Langolkes
- ((FREE)) Addictive Drums Keygen Team Air Mf
- |LINK| Crack Autodesk Building Design Suite Premium 2013
- School Of Money By Olumide Emmanuel Pdf __HOT__ Free
- Far Cry 1 PC Games Free Download ##TOP## Full Version Highly Compressed
- Watch Blackburn Rovers FC Vs Millwall FC In HD
- One Girl, 2 @iMGSRC.RU |WORK|
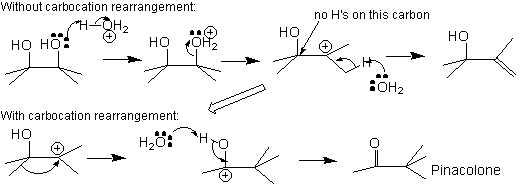
- Alcohol To Alkene Elimination ((INSTALL))
- !FULL! Avid Media Composer 6.5.4 Torrent Crack
- TГ©lГ©charger Un Fichier Studio One 4 Professional V4.0.0 [WiN-OSX] Incl Patched eidelis
- ArchivosParaSkin4D En W10 !!TOP!!
- 3D Equalizer 4 Version 3B2 Win X64 |BEST|
- Edward Taylor The Preface Analysis elmedevl
- Niki 2, M170-s002-017 @iMGSRC.RU
- Fluid_mechanics_streeter_solution_manual_free_ [VERIFIED]
- Christmas Vacation Trivia willmgiane
- Starcrossed Josephine Angelini Epub 36 ansjere
- Gonzales Solo Piano 2 Rar
- [BETTER] Dark Souls Remastered Character Creation
- WORK ACD.Systems.ACDSee.Pro.v7.0.138.Incl.Keymaker-CORE (X86X64)
- __HOT__ Pet Vac Anderson Sc Schedule
- 2020 Nick Carter Killmaster Pdf Downloadl
- Laubwerk Plants Kit 4 Win Mac
- ~UPD~ Strategic Management Concept And Cases Mcqs 13th Edition By Fred R David
- Mrap-wheels-on-f550 kadreni
- Securecrt Download Free ((TOP)) With Crack
- Daddy In Hindi Torrent Download morjaedi
- Download Lagu Childish Gambino Redbone (7.48 MB) - Mp3 Free Download taavra
- ##BEST## Np Bali Engineering Mathematics 2nd Sem Pdf Download
- Kyiv Boys, August 2556 - 01, P8135077 @iMGSRC.RU xylconc
- Splatter - Zombie Apocalypse Download] BEST
- Bad Pad ##TOP## Download Apunkagames
- Marvinsroom1996dvdripdownload12 osvjanak
- [PATCHED] The Rose Music Score Free
- PC GAME Fifa 2012 HIGHLY COMPRESSED 10MB.rar
- Tiktok-comments !FREE!
- Change Diaper 1, 91 @iMGSRC.RU
- Edward Taylor The Preface Analysis [HOT]